CMN Weekly (10 February 2023) - Your Weekly CRISPR Medicine News
By: Gorm Palmgren - Feb. 10, 2023
Top picks
- Researchers in Denmark and the US have explored the potential of a novel system for genome editing in human cells based on CRISPR-MAD7 - an alternative and royalty-free CRISPR-Cas12a nuclease developed from Eubacterium rectale (ErCas12a). Using a high-throughput engineering approach, the authors identified crRNAs that allow for ≤95% non-homologous end joining (NHEJ) and 66% frameshift mutations in various genes. They observed the high-cleavage fidelity of MAD7, resulting in an undetectable off-target activity. Moreover, they obtained ≤85% chimeric antigen receptor (CAR) insertions in primary T cells, thus exceeding the baseline integration efficiencies of therapeutically relevant transgenes using currently available virus-free technologies.
Research
- Chinese researchers have developed a rapid and portable hepatitis B virus (HBV) DNA detection assay for low-level viremia patients. The assay is based on CRISPR-Cas13a combined with recombinase-aided amplification (RAA) technology. The sensitivity, specificity, positive predictive agreement (PPA), and negative predictive agreement (NPA) values of dynamic plasma detection in patients on antiviral therapy were 100%, 92.15%, 93.75%, and 100%, respectively.
- A write-up of results from the phase 1 clinical trial of NTLA- 2001 for patients with transthyretin amyloidosis (ATTR) with cardiomyopathy is presented by MD Susy Kotit from the Aswan Heart Centre in Egypt, who was not herself involved in the study. Under the assumption that the data is correct and complete, the paper provides an excellent overview of the methodology and the results of this first-in-human in vivo CRISPR-Cas9 gene editing trial.
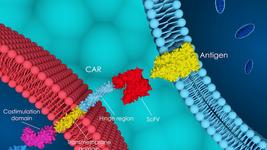
- Another write-up of results addresses the phase 1 clinical trial of gene-edited off-the-shelf CAR T cells to treat multiple myeloma. ALLO-715 is a first-in-class, allogeneic cell therapy developed by researchers at the Memorial Sloan Kettering Cancer Center and elsewhere.
Industry
- Ensoma reports that the Danish Business Authority has cleared its acquisition of Twelve Bio, according to Danish foreign direct investment laws. As a result, the Co-founder of Twelve Bio, Stefano Stella, has joined the leadership team at Ensoma as vice president of gene editing. Moreover, Guillermo Montoya, co-founder of Twelve Bio, and Shengdar Q. Tsai, a member of the scientific advisory board of Twelve Bio, will join the scientific advisory board at Ensoma.
- The first patient has been dosed in the phase 1 trial of Century Therapeutics gene-edited therapeutic CNTY-101 for relapsed or refractory CD19 positive B-cell lymphomas. CNTY-101 is an allogeneic cell therapy product candidate with six genetic modifications incorporated using sequential rounds of CRISPR. The modifications include a CD19-CAR, Allo-Evasion™ technology, IL-15 cytokine support and a safety switch.
- Fierce Pharma reports Vertex's launch plans for the first CRISPR gene editing therapy, exa-cel, to treat sickle cell disease and beta-thalassemia. Last month, the European Medicines Agency (EMA) validated a marketing authorisation application for exa-cel, but the therapy is still awaiting approval from the FDA.
Reviews
- Advances in gene therapy hold promise for treating hereditary hearing loss. The review addresses three main gene therapy strategies - gene replacement, gene suppression, and gene editing, summarising which approach is most appropriate for particular monogenic diseases based on different pathogenic mechanisms and then focusing on their successful applications for HHL in preclinical trials. Finally, it elaborates on the challenges and outlooks of gene therapy for HHL.
- CRISPR/Cas genome editing in triple negative breast cancer: Current situation and future directions. The review focuses on summarising the molecular subtypes of TNBC, the CRISPR system and its potential applications in TNBC treatment. Moreover, it discusses several emerging strategies for utilising the CRISPR-Cas system to aid in the precise diagnosis of TNBC and the limitations of the CRISPR-Cas system.
- Reversing the Central Dogma: RNA-guided control of DNA in epigenetics and genome editing. This review by researchers from Stanford University discusses how the normal flow of genetic information from DNA to RNA can be reversed by long noncoding RNAs (lncRNAs) and CRISPR, so the flow goes from RNA to DNA. The authors explore the parallels between these two fields and highlight opportunities for synergy and future breakthroughs.
- Application of CRISPR/Cas9 Technology in Cancer Treatment: A Future Direction. This review by researchers from Saudi Arabia examines CRISPR-Cas9 for tumour therapy research, which will help provide references for future studies on the pathogenesis of malignancy and its treatment.
News from CRISPR Medicine News
- On Monday, we brought a clinical roundup on gene-edited CAR T therapies. The roundup summarises the clinical-stage gene-edited CAR T candidates for cancer and looks at some of the significant edits they feature.
- We posted a clinical trial roundup on gene-editing therapies for renal cell carcinoma on Tuesday. Here, we summarise the two ongoing gene-editing clinical trials for renal cell carcinoma, the most common form of kidney cancer found in adults.
- Also, on Tuesday, we published a clinical update on a TALEN-edited CAR T-cell therapy for large B-cell lymphoma. The update looks at a phase 1/2 study for ALLO-501A, a TALEN-edited CAR T-cell therapy for large B-cell lymphoma.
To get more of the CRISPR Medicine News delivered to your inbox, sign up to the free weekly CMN Newsletter here.
Tags
CLINICAL TRIALS
IND Enabling
Phase I
Phase II
Phase III
IND Enabling
Phase I
Phase II
Phase III
Amyotrophic Lateral Sclerosis, ALS, or Frontotemporal Dementia FTD, (NCT04931862)
Sponsors:
Wave Life Sciences Ltd.
Sponsors:
Wave Life Sciences Ltd.
IND Enabling
Phase I
Phase II
Phase III