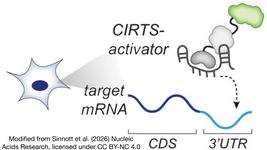
Relevant Interviews, Webinar and Trial Updates posted by CRISPR Medicine News.
Always stay updated with the free CMN Newsletter.
LogicBio Therapeutics
Main focus: Genomic medicines for genetic diseases
Company stage: Clinical
Diseases (gene editing): methylmalonic acidemia (MMA), Crigler-Najjar syndrome, tyrosinemia type 1, Wilson disease, others undisclosed
Genome editing tool: GeneRide
Funding stage: Public (NASDAQ:LOGC)
Location: Lexington, MA, USA
Website: https://www.logicbio.com/
Pipeline: https://www.logicbio.com/pipeline
Partners: Takeda Pharmaceuticals, Daiichi Sankyo, CANbridge Pharmaceuticals

LogicBio Therapeutics develops its GeneRide™ platform as a novel approch to gene editing. The technology harnesses the cells' natural DNA repair processes and does not require an externally supplied nuclease. The company currenly has one clinical development programme, targeting the rare disease methylmalonic acidemia. This candiate is currently being tested in a Phase I/II clinical trial.