CMN Weekly (16 June 2023) - Your Weekly CRISPR Medicine News
By: Karen O'Hanlon Cohrt - Jun. 16, 2023
Top picks
- Great Ormond Streeet Hospital made the news last December when it was announced that a young girl, Alyssa, with T-cell acute lymphoblastic leukaemia had been cured following treatment with an experimental base-edited T cell therapy. Alyssa was treated as part of a Phase 1 clinical trial, with engineered donor T cells that were transduced with lentivirus to express a CD7-targeting CAR, and she achieved molecular remission within 28 days after infusion of a single dose. Interim results from this trial were published yesterday in New England Journal of Medicine.
- The Road To CRISPR Vision. This contribution to Forbes, by President of ACCESS Health International William A. Haseltine, describes recent advances that use CRISPR to treat retinitis pigmentosa.
Research
- Scientists in the U.S. have developed a hepatocyte-specific base-editing therapeutic approach delivered by dual adeno-associated virus (AAV) to enable hepatocyte-specific targeting of the Angptl3 gene to lower blood lipid levels. Systemic AAV9-mediated delivery of AncBE4max, a cytosine base editor (CBE) targeting mouse Angptl3, resulted in the installation of a premature stop codon in Angptl3 with an average efficiency of 63.3 ± 2.3% in the bulk liver tissue. A near-complete knockout of the ANGPTL3 protein in the circulation was observed within 2-4 weeks following AAV administration. Furthermore, the serum levels of triglyceride (TG) and total cholesterol (TC) were decreased by approximately 58% and 61%, respectively, at 4 weeks after treatment. The findings, which were published yesterday in Cell and Bioscience, highlight the promise of liver-targeted Angptl3 base editing for blood lipid control, a strategy that is also being pursued by Verve Therapeutics (see more about Verve's VERVE-201 programme here).
- In an article published yesterday in Nature Biotechnology, a team of scientists in China and the U.S. reports a class of base editors that enable efficient adenine transversion, including precise A•T-to-C•G editing. They found that a fusion of mouse alkyladenine DNA glycosylase (mAAG) with nickase Cas9 and deaminase TadA-8e catalysed adenosine transversion in specific sequence contexts. Laboratory evolution of mAAG significantly increased A-to-C/T conversion efficiency up to 73% and expanded the targeting scope. Further engineering yielded adenine-to-cytosine base editors (ACBEs), including a high-accuracy ACBE-Q variant, that precisely install A-to-C transversions with minimal Cas9-independent off-targeting effects. ACBEs mediated high-efficiency installation or correction of five pathogenic mutations in mouse embryos and human cell lines. Founder mice showed 44-56% average A-to-C edits and allelic frequencies of up to 100%. The authors propose that adenosine transversion editors substantially expand the capabilities and possible applications of base-editing technology.
- In an article published this week in The Journal of Heart and Lung Transplantation, a team of scientists in Canada report the application of CRISPR-Cas epigenome editing to donor lungs to fine-tune immunomodulatory gene expression, exploring for the first time the therapeutic use of CRISPR-mediated transcriptional activation in the whole donor lung. They found that targeted transcriptional activation induced robust and titrable IL-10 upregulation in vitro, and that a combination of guide RNAs also allowed multiplex gene modulation of IL-10 and IL1 receptor antagonist. In vivo profiling demonstrated that adenoviral delivery of Cas9-based activators to the lung was feasible with the use of immunosuppression, which is routinely applied to organ transplant recipients. The transcriptionally modulated donor lungs retained IL-10 upregulation in isogeneic and allogeneic recipients. The findings suggest that transcriptional modulation of donor lungs via CRISPR may improve lung transplant outcomes by creating a more favourable immunomodulatory environment in the donor organ.
- Motivated to address limitations in currently available enzymes to modulate transcription, scientists at Epic Bio undertook a study that identified compact and potent activators from viral proteomes with robustness across silent and expressed genes in varied cell types using distinct catalytically-dead Cas (dCas) systems. By gathering insights from predicted 3D structures and machine learning models, the team could rationally engineer improved activators, both in potency and persistence, and they note that engineered activators achieved mitotically durable gene activation following transient delivery. The findings were shared this week in a manuscript on the pre-print server bioRxiv, and the authors propose that their discovery pipeline provides a predictive rubric for the systematic development of hypercompact activators from unannotated proteomes that broadly expand the epigenetic editing toolkit for therapeutic applications.
- Bacterial adaptive immune systems based on CRISPR-Cas maintain genome integrity and avoid autoimmunity by distinguishing between self and non-self, a process for which the CRISPR/Cas1-Cas2 integrase is required but not sufficient. In some microorganisms, the Cas4 endonuclease assists in CRISPR adaptation, but many CRISPR-Cas systems lack Cas4. In an article published earlier this week in Nature, a team led by Jennifer Doudna reports that an elegant alternative pathway in a type I-E system employs an internal DnaQ-like exonuclease to select and process DNA for integration using the protospacer adjacent motif (PAM). The natural Cas1-Cas2/exonuclease fusion (trimmer-integrase) catalyses coordinated DNA capture, trimming and integration. The team reports that five cryo-electron microscopy structures of the CRISPR trimmer-integrase, visualised both before and during DNA integration, show how asymmetric processing generates size-defined, PAM-containing substrates. Before genome integration, the PAM sequence is released by Cas1 and cleaved by the exonuclease, marking inserted DNA as self and preventing aberrant CRISPR targeting of the host. The authors propose that their findings support a model in which CRISPR systems that lack Cas4 use fused or recruited exonucleases for faithful acquisition of new CRISPR immune sequences.
Clinical
- Vertex Pharmaceuticals and CRISPR Therapeutics announced positive results from pivotal trials of exa-cel for transfusion-dependent beta thalassemia (BT) and severe sickle cell disease at the recent 2023 Annual European Hematology Association Congress. Both CLIMB-111 (for BT) and CLIMB-121 (for SCD) trials met their primary and key secondary endpoints at the pre-specified interim analysis for each trial, and all patients treated with exa-cel demonstrated clinical benefit, indicating the potentially transformative profile of exa-cel. The companies announced the same day that the FDA had accepted their Biologics License Applications for exa-cel for the treatment of severe SCD and transfusion-dependent BT.
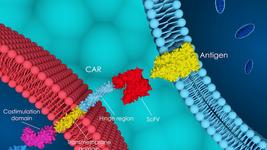
- Allogene Therapeutics provided additional ALLO-501/501A Phase 1 data in an oral presentation at the International Conference on Malignant Lymphoma (ICML), which was held this week in Switzerland. The data were obtained from the Phase 1 ALPHA/ALPHA2 trials of TALEN-edited ALLO-501/501A in 33 CAR T naïve patients with relapsed/refractory large B-cell lymphoma. Earlier in June, data from 12 patients, a subset of these 33 CAR T naïve patients, who received regimen being utilised in ongoing Phase 2 trials was presented at American Society of Clinical Oncology Annual Meeting. Across all 33 patients, the complete response (CR) rate was 42% with 30% maintaining a CR at Month 6. Median duration of response for both the overall population (n=33) and the patients treated with the Phase 2 regimen (n=12) was 23.1 months. See the official press release for more details. You can learn more about ALLO-501/501A in a previous clinical trial update here.
- This week, Intellia Therapeutics announced new positive clinical data from the ongoing Phase 1 trial of NTLA-2002 in hereditary angioedema (HAE). NTLA-2002 is an in vivo CRISPR medicine candidate being developed as a single-dose treatment for HAE. The data, with a cut-off date of February 17, 2023, were shared in a late-breaking presentation at the European Academy of Allergy and Clinical Immunology Hybrid Congress 2023, which was held last week in Germany, and virtually. The data revealed that across all 10 patients included for analysis, a single dose of NTLA-2002 led to a 95% mean reduction in monthly HAE attack rate through the latest follow-up. Learn more about NTLA-2002 and HAE in a previous clinical trial update here.
Industry
- Yesterday, Verve Therapeutics announced an exclusive global collaboration with Eli Lilly to advance one of Verve’s in vivo base-editing programmes for the treatment of atherosclerotic cardiovascular disease. The collaboration will see the two companies focus on Verve's pre-clinical programme targeting lipoprotein(a) (Lp(a)). Elevated Lp(a) is an established and genetically validated, independent risk factor for atherosclerotic cardiovascular disease, ischemic stroke, thrombosis, and aortic stenosis. Under the terms of the collaboration, Verve will advance the R&D of the Lp(a) programme through the completion of Phase 1 clinical development. Lilly will be responsible for subsequent development, manufacturing, and commercialisation of the Lp(a) programme. See the official press release for further details.
- Editas Medicine announced this week that it intends to offer and sell $125 million of shares of its common stock in an underwritten public offering. Editas Medicine intends to grant the underwriters a 30-day option to purchase up to an additional $18.8 million of shares of its common stock. All of the shares in the offering are to be sold by Editas Medicine. Details of the offering as well as pricing of the stocks can be found here and here.
- Future Fields and Jenthera Use Fruit Flies for In Vivo CRISPR Therapy. Future Fields, a biomanufacturing company based in Canada, is using whole fruit flies to produce recombinant proteins for in vivo CRISPR-based gene therapy developed by pre-clinical Canadian biotech company Jenthera Therapeutics in a sustainable and cost-efficient manner. The two companies have announced a collaboration to use Future Fields’ EntoEngine™, the world’s first synthetic biology system that uses fruit flies for recombinant protein production, to develop a first-of-its-kind cancer-fighting protein. Jenthera Therapeutics’ hybrid molecules are made up of cell-specific nano-antibody-binders (NABs) and CRISPR ribonucleoproteins (RNPs).
Reviews
- An Expert Overview on Therapies in Non-Transfusion-Dependent Thalassemia: Classical to Cutting Edge in Treatment. This review provides an overview of functional mechanisms, advantages, and disadvantages of the classic to latest new treatments for non-transfusion-dependent thalassemia patients, depending on the disease severity divided into the typical management strategies for patients with β-TI such as foetal haemoglobin induction, splenectomy, bone marrow transplantation, transfusion therapy, and herbal and chemical iron chelators.
- Repurposing prokaryotic clustered regularly interspaced short palindromic repeats-Cas adaptive immune system to combat antimicrobial resistance. One of the most urgent challenges facing public health is the rise in infections by antibiotic-resistant microorganisms. CRISPR-Cas has gained considerable attention as an alternative antibacterial therapy. This review deals with the development of CRISPR-Cas antimicrobials and their delivery challenges.
- Base Editing and Prime Editing: Potential Therapeutic Options for Rare and Common Diseases. Authors at Perelman School of Medicine at the University of Pennsylvania provide an overview of base editors (BE) and prime editors (PE), including their structures, mechanisms, and differences from standard CRISPR-Cas9 genome editing. They describe several examples of the use of BE and PE to improve rare and common disease phenotypes in pre-clinical models and human patients, with an emphasis on in vivo editing efficacy, safety, and delivery method. The authors also discuss recently developed delivery methods for BE and PE that may be used in future clinical settings.
- Advancements of in vitro transcribed mRNA (IVT mRNA) to enable translation into the clinics. This review describes the advances that have supported the adoption of in vitro-transcribed mRNA in the clinics, including optimisation of the IVT mRNA structural components, synthesis, and lastly concluding with different classes of IVT RNA. The authors foresee that a continuing interest in driving IVT mRNA technology will enable a safer and more efficacious therapeutic modality to address emerging and existing diseases.
- Evolution of Nanomedicine Formulations for Targeted Delivery and Controlled Release. In this review article, scientists from AstraZeneca review historical nanomedicine platforms, characterisation methodologies, challenges to their clinical translation, and key quality attributes for commercial translation with a view to their developability into a genomic medicine. New nanoparticle systems for immune targeting, as well as in vivo gene editing and in situ CAR therapy, are also highlighted as emerging areas.
News from CRISPR Medicine News
- On Wednesday, we published a clinical trial update, with news from Editas Medicine. The company recently announced positive initial safety and efficacy data for its CRISPR-edited cell therapy EDIT-301. The data was obtained from the first four sickle cell disease patients and the first beta-thalassemia patient treated in the ongoing RUBY and EdiTHAL clinical trials. Read the full update here.
Huh, Heh, Wow
- Scientists create an avocado that doesn't turn black after being cut. By using CRISPR to disrupt the gene that drives oxidation after an avocado is cut and its inside comes into contact with the air, Chile's most-consumed fruit will last longer in homes without turning black. Read the full story here.
To get more of the CRISPR Medicine News delivered to your inbox, sign up to the free weekly CMN Newsletter here.
Tags
CLINICAL TRIALS
IND Enabling
Phase I
Phase II
Phase III
IND Enabling
Phase I
Phase II
Phase III
Amyotrophic Lateral Sclerosis, ALS, or Frontotemporal Dementia FTD, (NCT04931862)
Sponsors:
Wave Life Sciences Ltd.
Sponsors:
Wave Life Sciences Ltd.
IND Enabling
Phase I
Phase II
Phase III