CRISPR-Armed Phages Clear First Human Safety Bar
CMN Intelligence - The World’s Most Comprehensive Intelligence Platform for CRISPR-Genomic Medicine and Gene-Editing Clinical Development
Providing market intelligence, data infrastructure, analytics, and reporting services for the global gene-editing sector. Read more...

The Copenhagen-based biotech has spent the past several years engineering bacteriophages to precisely target Escherichia coli in the gastrointestinal tract, working to prevent bloodstream infections in patients with haematological malignancies undergoing stem-cell transplantation. The newly published first-in-human trial now provides the safety and feasibility signal required to move the programme into patient studies.
For Eric van der Helm, VP Business Development at SNIPR, the significance goes beyond the dataset itself. »This study delivered a very clear message on feasibility and safety. In a randomised, controlled first-in-human trial, we showed that orally administered engineered phages are safe and well-tolerated, including at higher doses. This has not been shown before for an orally dosed CRISPR–Cas-armed phage therapeutic, and it sets an important benchmark for the field.«
SNIPR arms phages with programmable killing
The scientific groundwork for SNIPR001 was published in Nature Biotechnology in 2024, describing a development process that builds on classic phage therapy, layering programmable killing on top. After screening 162 wild-type lytic phages for breadth and complementarity, SNIPR selected candidates that bind different bacterial surface receptors, reducing the chance that E. coli could evade the cocktail through a single mutation.
“We position SNIPR001 primarily as a way to prevent infections from happening in the first place – and we have encouraging data supporting the potential for treating active infection as well”Christian Grøndahl, Founder and CEO at SNIPR
What makes SNIPR001 distinct is the CRISPR-Cas arming, which adds an intracellular lethality layer to the phage's normal lytic cycle. Once inside E. coli, the phage expresses the CRISPR machinery, and the Cas3 nuclease degrades the bacterial genome at pre-programmed sites – ensuring cell death even when lysis alone would not finish the job. By targeting multiple critical genomic regions at once, the system makes it considerably harder for the bacterium to survive through mutation. In laboratory tests, the CRISPR–Cas-armed phages generated fewer resistant survivors than wild-type comparators – a durability signal that matters commercially as much as initial potency.
The overall approach – combining complementary receptor targeting, a modular Cas3 killing system and a development process focused on stability and manufacturability – is likely to strike investors as a true platform rather than a one-off asset.
The Phase 1 trial enrolled 36 healthy volunteers who received escalating oral doses of 10⁸, 10¹⁰ and 10¹² plaque-forming units, taken twice daily for seven days. The study was randomised, double-blind and placebo-controlled. No serious adverse events were reported, and overall adverse event rates were similar to those seen with placebo.
Patient trial targets a higher-burden microbiome
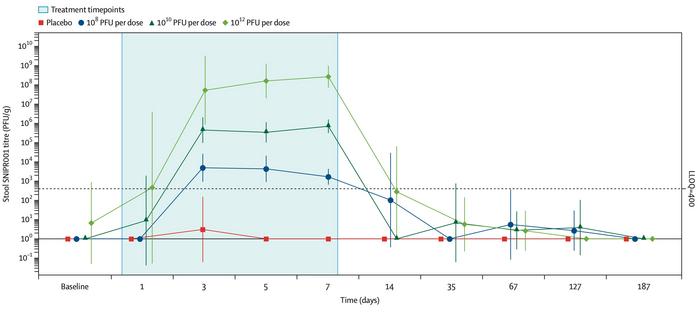
SNIPR001 was recovered from stool in dose-proportional amounts, and no systemic exposure was detected in plasma or urine (see Figure 1). Gut microbiome composition was unchanged throughout, and SNIPR001 remained undetectable six months after the final dose. This indicates a pharmacokinetic footprint consistent with regulatory expectations for live biotherapeutics.
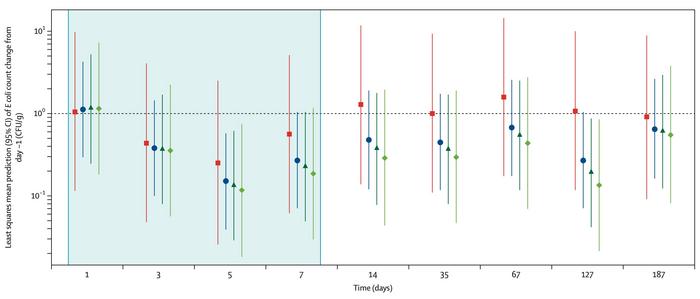
Efficacy was not the primary endpoint. Nevertheless, the highest-dose group showed a 78% reduction in E. coli levels versus placebo at day 14, though this did not reach statistical significance (see Figure 2). Van der Helm is explicit about how the company reads that signal. »It's important to frame the result in the context of the study design: this trial was powered for safety, not for detecting statistically significant reductions in E. coli. So we did not anticipate a conclusive efficacy readout. That said, the fact that we saw a dose-dependent trend is meaningful to us; it suggests E. coli target engagement and supports the decision to move forward into a phase 1b/2a trial in patients.«
SNIPR001 is built around a clearly defined, high-risk group: patients with haematological cancer undergoing stem-cell transplantation, where neutropenia and damage to the gut lining increase the risk of bloodstream infections, and fluoroquinolone resistance is common. The ongoing phase 1b/2a trial, now more than halfway enrolled across eight US cancer centres, is evaluating SNIPR001 in precisely this population.
»The biology is different,« says Christian Grøndahl, Founder and CEO at SNIPR. »The microbiome of the intended patient population can differ substantially from that of healthy volunteers, and E. coli can be present at much higher abundance. In that context, we expect SNIPR001 to have more dynamic range to demonstrate activity.« A dysbiotic, high-burden microbiome may yield a clearer therapeutic window – and a more compelling efficacy signal – than a stable, healthy gut.
Platform strategy targets a structurally broken market
CRISPR-based antimicrobials remain an emerging category. SNIPR001 sits between traditional antibiotics and broad microbiome therapeutics: a genetically programmed, pathogen-specific antibacterial with a defined molecular mechanism. The wider market context is well established – in 2019, antibiotic-resistant bacteria were estimated to have directly caused 1.27 million deaths globally, with E. coli the single largest contributor, and few novel antibiotic classes are advancing through late-stage development.
Grøndahl frames the positioning in systemic terms. »Antibiotic resistance continues to rise globally, and antibiotics remain a mainstay of modern medicine – without effective antibiotics, many routine hospital procedures become far more difficult if not impossible. We therefore believe innovation in antimicrobials is essential to protect both current and future healthcare. In that landscape, we position SNIPR001 primarily as a means to prevent infections in the first place, particularly in high-risk settings. At the same time, we have encouraging data supporting the potential for treating active infection. Strategically, it's also important that SNIPR001 is built on a platform: we've applied the same approach used to generate SNIPR001 to create CRISPR-based medicines against other high-priority threats, including Klebsiella and Pseudomonas.«
“We showed that orally dosed engineered phages are safe and well tolerated – this has not been shown before, and it sets an important benchmark for the field”Eric van der Helm, VP Business Development at SNIPR
The field is still taking shape. Locus Biosciences has published phase 2 safety and pharmacodynamic data for LBP-EC01, a CRISPR-Cas3-enhanced phage cocktail targeting E. coli in urinary tract infections, while Seres Therapeutics' SER-155 – a bacterial consortium therapeutic aimed at bloodstream infections and graft-versus-host disease in allogeneic HCT – has concluded it’s phase 1b evaluation. The indications differ, but all three programmes share a common premise: that targeted microbiome intervention can do what broad-spectrum antibiotics cannot in high-risk hospital populations.
SNIPR001 holds an FDA fast-track designation for the prophylaxis of bloodstream E. coli infections in patients with haematological malignancy at risk of neutropenia. The therapy is still experimental, and phase 1 data in healthy volunteers are only the beginning. But they mark a real step forward – from preclinical promise to a therapy that has now cleared its first human test. Whether it can reduce infection rates in the patients who need it most is the question the ongoing trials will have to answer.
The phase 1 clinical results were published in The Lancet Microbe on 3 March 2026 (Safety, recovery, and pharmacodynamics of CRISPR–Cas therapeutic SNIPR001: a phase 1, randomised, double-blind, first-in-human, dose-escalation study). The preclinical characterisation of SNIPR001 was published in Nature Biotechnology in 2024 (Engineered phage with antibacterial CRISPR–Cas selectively reduce E. coli burden in mice).
To get more CRISPR Medicine News delivered to your inbox, sign up to the free weekly CMN Newsletter here.
Tags
ArticleInterviewNewsHaematological malignancies (blood cancers)Cas3SNIPR BiomeClinical
CLINICAL TRIALS
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.