Scribe Therapeutics Positions Epigenetic CRISPR for Durable LDL-C Reduction
CMN Intelligence - The World’s Most Comprehensive Intelligence Platform for CRISPR-Genomic Medicine and Gene-Editing Clinical Development
Providing market intelligence, data infrastructure, analytics, and reporting services for the global gene-editing sector. Read more...

The announcement followed the January 2026 J.P. Morgan Healthcare Conference – a venue as much about capital allocation and strategic partnerships as scientific validation. The timing signals that Scribe is pitching not only to the scientific community but also to investors and potential collaborators as it attempts to translate CRISPR technology into a common-disease business model.
At a strategic level, Scribe is testing whether CRISPR-based medicines can move beyond rare-disease economics and into a cardiometabolic indication with patients measured in the hundreds of millions. That transition demands more than molecular precision. It requires durability, manufacturing discipline and pricing logic compatible with population-scale medicine.
For chief executive Benjamin Oakes, the commercial argument begins with a structural mismatch.
»If you look at the LDL-C lowering space, there are already many therapies available,« he says. »The fundamental issue is that none of the currently approved therapies matches the durability of effect to the duration of the disease.«
“If we can develop a therapy that does not have to be permanent but is highly durable… we could potentially provide an end-state solution for LDL-C lowering”Benjamin Oakes, CEO of Scribe Therapeutics
Atherosclerotic cardiovascular disease unfolds over decades. Yet most pharmacological interventions rely on continuous dosing, often for life. In practice, that model proves difficult to sustain.
Real-world adherence to lipid-lowering therapies declines over time, even for relatively infrequent dosing regimens, and more so for daily oral treatments such as statins.
The economic implication is clear: even highly effective drugs underperform at the population level if patients do not persist. Durability, in this framing, becomes a lever not only on biology but on health system efficiency.
From cutting genes to changing their activity
STX-1150 works by turning down the liver’s production of PCSK9 using a modified CRISPR-CasX system that has been engineered not to cut DNA. The therapy is delivered in vivo via lipid nanoparticles that carry the molecular instructions into liver cells. Once there, Scribe’s Epigenetic Long-Term X-Repressor – or ELXR – places repressive chemical marks at the PCSK9 gene, effectively switching it off without changing the underlying DNA sequence. Unlike nuclease-based editing or base editing approaches, the therapy is designed to avoid permanent genomic changes.
In a recent preprint, Scribe scientists describe how they built an allosterically regulated version of the DNA methyltransferase DNMT3A into a catalytically inactive version of an engineered CasX, reconstructing the DNMT3A enzyme’s native autoinhibitory control loop (see Figure 1). In cellular and animal models, this additional regulatory layer reduced transcriptome-wide off-target perturbations by 10- to 100-fold compared with non-allosteric constructs, while maintaining or enhancing on-target repression.
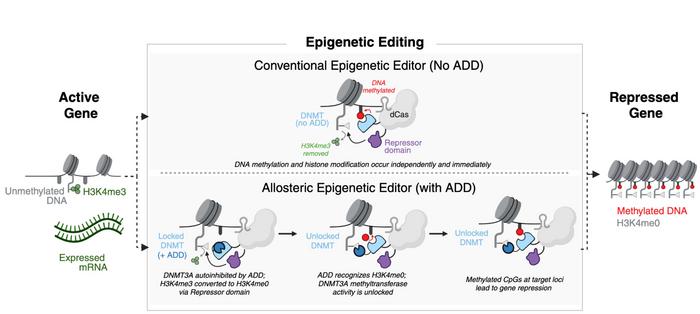
Oakes perceives epigenetic silencing as a pragmatic evolution of mRNA-silencing technologies. Because epigenetic silencing does not permanently alter the genome, the approach should offer some of the conceptual advantages of such mRNA-silencing technologies while aiming for greater durability, potentially allowing a single administration to last for years before redosing is needed.
The company’s corporate pedigree reinforces that positioning. Co-founded by Nobel Prize winner Jennifer Doudna and backed by leading life sciences investors, Scribe has formed strategic collaborations with Sanofi and Eli Lilly. Those alliances provide external validation of the underlying engineering platform, even as STX-1150 becomes the first test of its scalability in a high-prevalence indication.
Durability as a commercial lever
An abstract presented at the American Heart Association’s 2025 Scientific Sessions adds granularity to the durability claims. In primary human hepatocytes in vitro, a prototype of STX-1150 achieved more than a 95% reduction in secreted PCSK9. In transgenic mice expressing human PCSK9, a single dose reduced circulating protein levels by more than 95%, sustained for over 310 days.
In non-human primates, a single intravenous dose of 0.75 mg/kg lowered LDL-C by at least 50%, with the effect lasting nearly 18 months and ongoing and liver enzyme levels remaining comparable to those of controls. When the same dose was given more than once, the reductions deepened in a stepwise manner, resulting in sustained LDL-C lowering of approximately 40% to 60% over 180 days.
Taken together, the data support the company’s claim that STX-1150 can deliver durable and adjustable lipid-lowering at sub-1 mg/kg doses, without permanent genomic modification.
For a common disease application, titratability may prove commercially significant. It suggests a therapy that can be calibrated rather than positioned as a binary, irreversible intervention – potentially easing both regulatory and reimbursement discussions.
»If we can develop a therapy that does not have to be permanent but is highly durable – durability measured in years to decades – we could potentially provide an end-state solution for LDL-C lowering,« Oakes says. The financial question follows immediately. »If you offer a decade of durability, who pays for that decade upfront?«
“With a durable in vivo therapy, once a patient is treated, you move forward. That changes the scalability equation entirely”Benjamin Oakes, CEO of Scribe Therapeutics
That question sits at the centre of the cardiometabolic gene medicine debate. Ultra-rare therapies tolerate high upfront pricing because patient numbers are small. A preventive intervention targeting millions must operate under different assumptions.
The logic is pharmacoeconomic rather than aspirational. A payer currently spending thousands of dollars annually for decades on combination lipid-lowering therapy could rationalise a single intervention representing a fraction of cumulative lifetime cost – particularly if it removes adherence variability from the equation.
Engineering for scale
Scalability is where the real test begins. By some estimates, around 200 million people across the US and EU have high cholesterol that should be treated, and roughly three-quarters of them are not at target. A market of that size requires manufacturing economics more akin to large-scale vaccine production than to bespoke cell therapy.
STX-1150 is delivered in vivo via lipid nanoparticles that encapsulate mRNA and a guide RNA. »We still have to manufacture mRNA and guide RNA at large scale,« Oakes says. »But it is an engineering challenge, not a biological limitation. And engineering challenges can be solved.«
A durable therapy also alters capacity dynamics. »With a durable in vivo therapy, once a patient is treated, you move forward. That changes the scalability equation entirely,« he adds. Unlike chronic injectables, manufacturing throughput is not consumed by re-treating the same cohort year after year.
If its allosterically regulated CasX-based epigenetic editor can prove itself in a large, high-prevalence indication, it would not only de-risk STX-1150. It would establish a scalable template for tackling other cardiometabolic targets without committing to permanent genome alteration.
For now, the next inflection point is clinical. Scribe expects to initiate a first-in-human Phase 1 study in individuals with hypercholesterolaemia at elevated cardiovascular risk in mid-2026. While the trial is designed to evaluate safety and tolerability as the primary objective, it will also assess the levels of LDL-C lowering to see how the reduction previously observed in non-human primates translates to humans.
If it does, STX-1150 may offer something the lipid-lowering field has long sought – a therapy whose duration of effect finally matches the duration of the disease.
This article draws on an interview with Benjamin Oakes, a Scribe Therapeutics press release from 20 January 2026, a preprint on bioRxiv from 14 January 2026, and an abstract from the American Heart Association's 2025 Scientific Sessions from 3 November 2025.
To get more CRISPR Medicine News delivered to your inbox, sign up to the free weekly CMN Newsletter here.
Tags
ArticleInterviewNewsIn vivoAtherosclerotic heart diseaseEpigenome editing (e-GE)CasXScribe TherapeuticsPre-clinical
CLINICAL TRIALS
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.