In situ sequencing maps in vivo editing
CMN Intelligence - The World’s Most Comprehensive Intelligence Platform for CRISPR-Genomic Medicine and Gene-Editing Clinical Development
Providing market intelligence, data infrastructure, analytics, and reporting services for the global gene-editing sector. Read more...
Traditional assessments of in vivo gene editing rely on next-generation sequencing of homogenised tissue samples, which provides average editing rates but obscures important spatial variation across different cell types and tissue regions.
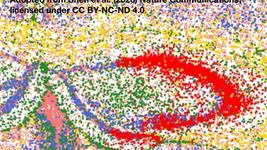
The research team at University of Zurich established imaging-based in situ sequencing (ISS) to overcome this limitation, enabling direct visualisation of editing events within intact tissues at single-cell resolution. The technology uses padlock probes that specifically recognise edited or unedited sequences, followed by rolling circle amplification and sequencing-by-synthesis to map editing outcomes whilst preserving tissue architecture.
Initial validation in cultured cells demonstrated that ISS accurately detected both adenine base editing and prime editing events, with editing rates closely matching fluorescent reporter expression. When applied to mouse brain tissue treated with adeno-associated viral vectors encoding intein-split base editors, ISS revealed substantially higher editing rates in cortical neurons compared to astrocytes, and showed that most edited cells carried modifications on both gene copies rather than single alleles.
The technique proved equally effective for mapping base editing in liver tissue, where researchers used metabolic zone markers to demonstrate widespread editing across periportal, intermediate, and pericentral hepatocyte populations following both AAV and RNA-lipid nanoparticle delivery. Notably, repeated dosing experiments in cynomolgus macaques showed that initial RNA-LNP treatment did not affect the efficiency or distribution of subsequent editing, supporting the feasibility of multiple therapeutic interventions.
The study was led by Sharan Janjuha, Tatjana Haenggi and Gerald Schwank at the University of Zurich, Switzerland. It was published in Nature Biomedical Engineering on 19 September 2025.
To get more CRISPR Medicine News delivered to your inbox, sign up to the free weekly CMN Newsletter here.
Tags
ArticleCMN BriefsNewsIn vivoRibonucleoprotein (RNP)Adeno-associated virus (AAV)Next gen sequencingBase editorsCas9Prime editors
CLINICAL TRIALS
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.