INSTALL Makes Non-Viral Gene Insertion Work In Vivo
CMN Intelligence - The World’s Most Comprehensive Intelligence Platform for CRISPR-Genomic Medicine and Gene-Editing Clinical Development
Providing market intelligence, data infrastructure, analytics, and reporting services for the global gene-editing sector. Read more...

For Benjamin Kleinstiver, Associate Professor at Harvard Medical School and senior author of the new study, the strategic significance is inseparable from a structural problem in the way the genetic medicines industry currently operates. Most inherited diseases are not caused by a single shared mutation – they are caused by dozens, hundreds, sometimes thousands of distinct variants, each calling for, in principle, its own bespoke therapeutic.
»In the context of new classes of genetic therapies, this approach supports the continued development of integration methods that overcome the heterogeneity of patient mutations,« Kleinstiver tells CRISPR Medicine News, »motivating the continued development of single mutation-agnostic therapies capable of treating patients regardless of their specific underlying genetic defect.«
Developed at Massachusetts General Hospital in collaboration with Full Circles Therapeutics and other laboratories across Mass General Brigham, INSTALL reflects a broader effort to make large-scale, non-viral gene insertion workable in clinically relevant settings.
The DNA donor becomes the bottleneck
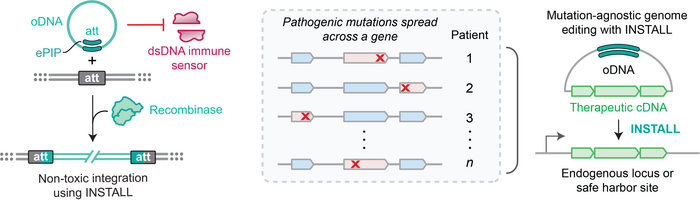
That ambition has long been the promise of gene insertion: deliver a functional copy of the entire gene rather than correcting each individual mutation. One obstacle has been delivery.
Viral vectors – above all, adeno-associated virus (AAV) – have dominated the field commercially, but their limitations are well known: high manufacturing costs, pre-existing immunity in a significant share of patients, and an inability to redose once the body has mounted an immune response to the capsid. Lipid nanoparticles sidestep many of those problems but deposit their contents into the cytoplasm, where double-stranded DNA is immediately recognised as foreign material and triggers inflammation.
“Mice treated with INSTALL were nearly indistinguishable from PBS-treated or mRNA-treated controls in terms of viability and behavior; conversely, dsDNA-treated animals rapidly developed fatigue and fatal immune responses”Connor Tou
Recombinases – enzymes that can insert gene-sized sequences at defined genomic addresses while minimising exposed DNA double-strand breaks, and thereby limiting associated toxicity and unintended genomic consequences – have attracted growing commercial interest as a route around AAV's limitations. Companies like Seamless Therapeutics and Stylus Medicine have invested substantially in engineering recombinases to act at therapeutically relevant sites. But the DNA donor itself has received comparatively little attention.
»In conventional approaches, recombinases must be paired with large double-stranded DNA (dsDNA) donor molecules carrying the cargo-of-interest,« explains Connor Tou, PhD, in the Kleinstiver lab at MGH and co-first and co-corresponding author of the paper. »However, the dsDNA donors can be detected in the cytosol of mammalian cells by various immune sensors, resulting in a strong DNA innate immune response that leads to cellular toxicity, disruption of gene expression, and in animals, fatal systemic inflammation.«
The breakthrough behind INSTALL came from looking at how nature had already solved an analogous problem. Many bacteriophages carry circular single-stranded DNA (cssDNA) genomes and have evolved mechanisms to integrate them into bacterial chromosomes, with the cell's own polymerases synthesising the complementary strand after the fact. Single-stranded DNA largely evades the sensors that trigger innate immunity: for example, cGAS, which initiates the STING inflammatory cascade, has more than ten times weaker affinity for single-stranded DNA than for double-stranded DNA.
»The 'aha' moment happened when we realized that across evolution, many phage and bacterial processes naturally support DNA integration reactions when using circular single stranded DNA (cssDNA) substrates with recombinases,« Tou tells. »By engineering and recapitulating these natural mechanisms but now in mammalian cells, we developed INSTALL.«
In its most refined form, INSTALL-2e, the platform pre-anneals the cssDNA donor to a short chemically modified oligonucleotide that reconstitutes just enough double-stranded DNA for the recombinase to recognise, while keeping the overall donor below the detection threshold of innate immune sensors. The result is a molecule that may also benefit from the structural flexibility of single-stranded DNA to facilitate nuclear import.
INSTALL clears the in vivo barrier
In primary human T cells, INSTALL-2e achieved approximately four times higher integration efficiency than double-stranded DNA, and accommodated cargos of 6 kb or larger – beyond the carrying capacity of AAV. As a therapeutic proof-of-concept, a 4.8 kb copy of ABCD1, the gene mutated in X-linked adrenoleukodystrophy, was integrated and shown to produce a functional downstream protein.
The in vivo results may be commercially the most consequential. At equivalent doses delivered via lipid nanoparticles, double-stranded DNA killed all treated mice within one to three days. INSTALL-2e at the same or higher doses produced no observable adverse effects.
»The results from our in vivo experiments were particularly striking: mice treated with INSTALL were nearly indistinguishable from PBS-treated or mRNA-treated controls in terms of viability and behavior; conversely, dsDNA-treated animals rapidly developed fatigue and fatal immune responses,« Tou says.
Because INSTALL is non-viral, redosing is also feasible – impossible with AAV – and a two-dose protocol achieved approximately 1% bulk liver integration, with genome-wide sequencing attributing reads only to the on-target site and detecting no or very-low-level integration at previously nominated off-target loci.
The study was a large collaborative effort. Key intellectual and experimental contributions came from Howard Wu and Keqiang Xie of Full Circles Therapeutics, a company specialising in the manufacturing and commercialization of circular single-stranded DNA. The laboratory of Patricia Musolino at the MGB Center for Genomic Medicine led integration and immunology studies in mice, while the laboratory of Natalie Artzi at the MGB Gene and Cell Therapy Institute designed and formulated the LNP delivery vehicles.
“We are optimistic that non-viral gene insertion with INSTALL will facilitate the development of mutation-agnostic genetic therapies”Benjamin Kleinstiver
The remaining engineering work is substantial. In vivo integration efficiencies of around 1% are unlikely to be sufficient for therapeutic effect in most target diseases, and Tou and Kleinstiver are candid about the roadmap:
»A primary focus will be identifying LNP formulations capable of efficiently co-delivering recombinase mRNA and INSTALL oDNA donors. Concurrently, refining the chemical modifications and sequence composition of the PIP oligo that is annealed to the cssDNA donor should maximize integration efficiencies by improving stability and resistance to degradation. Furthermore, the continued engineering of recombinases to reprogram them to directly integrate into therapeutic loci within the human genome would simplify INSTALL approaches and enhance overall translation potential.«
The analogy the authors draw is instructive: the chemical modifications that neutralised the immunogenicity of siRNA and mRNA took years to optimise but ultimately unlocked two entirely new classes of approved medicines.
»We are optimistic that non-viral gene insertion with INSTALL will facilitate the development of mutation-agnostic genetic therapies,« Kleinstiver concludes.
The original article "Immune evasive DNA donors and recombinases license kilobase-scale writing" was published today, 11 March 2026, in Nature.
To get more CRISPR Medicine News delivered to your inbox, sign up to the free weekly CMN Newsletter here.
Tags
ArticleInterviewNewsIn vivoLipid-based nanoparticleFull Circles TherapeuticsPre-clinical
CLINICAL TRIALS
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.
Sponsors:
Base Therapeutics (Shanghai) Co., Ltd.







