2020 was an incredible CRISPR year

It's been an incredible year for the CRISPR genome editing tool developed only eight years ago. This year CRISPR was awarded the Nobel Prize in Chemistry, entered the human body, and saw victories in clinical trials that it is no longer just a dream to eliminate hereditary diseases but reality. In 2020 CRISPR truly brought medicine into a new era of gene editing - here are our five highlights of 2020.
Nobel Prize in Chemistry
Its been clear for several years that it was not so much a question if the CRISPR-Cas9 gene-editing would be awarded a Nobel Prize as when. This year the committee lifted the veil and awarded the Nobel Prize in Chemistry to Emmanuelle Charpentier and Jennifer Doudna for their pioneering work.
Just eight years after their groundbreaking paper CRISPR has conquered molecular biology and literally changed the daily work in molecular biology labs around the world.
Charpentier and Doudna successfully developed the prokaryotic CRISPR immune system into a simple easy-to-use programmable gene-editing tool with a guide RNA molecule that directs the Cas9 endonuclease from Streptococcus pyogenes to cleave sequence-specific regions of DNA.
The CRISPR-Cas9 technology proved to be a much simpler and more versatile method for editing the genomes of living organisms than earlier technologies such as zinc-finger nucleases and TALENs. And quickly found use in everything from fundamental research such as creating unique model organisms over creating new disease-resistant crops to developing potential cures for genetic diseases.
It is undoubtedly clear that discovering the 'genetic scissors' and improved ability to edit genomes is a monumental achievement and well worth celebrating. CRISPR's contributions to science will continue and so will gene editings impact on society in general. There is a huge potential to be realized in the years to come, and with the first real cures already taking shape, we may already see another CRISPR Nobel Prize in Medicine on the horizon.
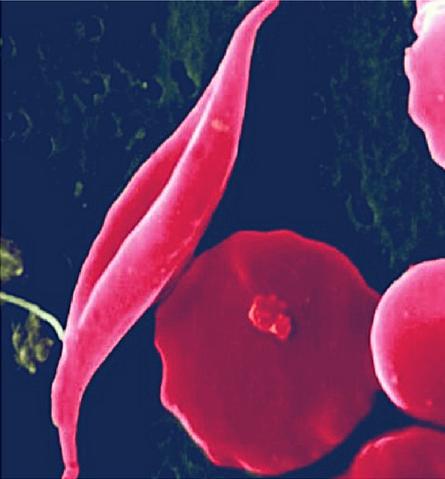
Victory at the clinical trials
The hype surrounding CRISPR is certainly high, but this year results from ongoing clinical trials is living up to the hype. Data presented in June and December 2020 caused a lot of excitement in the CRISPR community: A one-time cure for the two inherited blood disorders - Sickle cell disease and β-thalassemia.
The therapy called CTX001 is an ex vivo cell therapy that collects the patients own haematopoietic stem cells and use CRISPR-Cas9 to make a small specific deletion that results in reactivating a fetal version of haemoglobin (Hbf). When reintroduced to the patients, the edited stem cells establish a permanent supply of healthy red blood cells producing Hbf.
So far, the data show consistent and sustained health improvements for a handful of patients with the first patient, Victoria Gray, receiving the treatment 17 months prior. In essence, the data amounts to a functional cure for the two genetic diseases.
The companies developing the therapy, Vertex Pharmaceuticals and CRISPR Therapeutics, are quickly moving forward with more patients to get enough data for approval of the potentially curative treatment.
First CRISPR gene editing in vivo
2020 also marked the first time CRISPR gene editing was performed directly in the human body. In March, the first CRISPR therapy was injected in the eye. Then in November, a different treatment was injected systemically in a patient.
Editas Medicine and Allergan injected their CRISPR-Cas9 therapy, EDIT-101, directly into the eye of a patient to treat congenital blindness caused by Leber's congenital amaurosis type 10. Since the first adult cohort has been completed, we are looking forward to the first data being released.
In November, Intellia Therapeutics took in vivogene editing one step further by delivering their CRISPR-based therapy systemically in the bloodstream of a patient to treat hereditary transthyretin amyloidosis (hATTR) with polyneuropathy. Intellia's therapy, NTLA-2001, is delivered via lipid nanoparticles designed to target the liver specifically. The CRISPR-agent will knock out the mutated TTR gene and stop a buildup of the misfolded TTR protein. The therapy can potentially cure the patients with a single course of treatment, but so far it is a phase 1 study for safety and dose-finding and will enrol as many as 38 participants.

Cancer CRISPR therapy works without serious side-effects
The list of new gene-editing clinical trials is growing fast. But in 2020 pioneering CRISPR cancer therapies stood out with results published in peer-reviewed scientific journals. Both clinical trials were CRISPR gene-editing versions of immunotherapy - instead of blocking the immune 'safety switch' called PD1 with antibodies; they use CRISPR to remove PD1 from the patients own T cells ex vivo. The T cells were then re-infused in patients with advanced cancers, and both trials showed the feasibility and positive safety data, if not much efficiency.
The first study was led by immunotherapy pioneer Carl June, University of Pennsylvania, US, directed the T cells to attack tumours in three patients with advanced refractory and metastatic cancer.
The second was a Chinese study that began in 2016 and enrolled 22 patients to treat aggressive lung cancer.
The therapies did not go as well as hoped for the patients - most patients died, and the few still alive were put on different treatments. However, both studies were safety and feasibility studies - the primary objective was not to cure the disease - and by that measure, both are important 'first in human' milestones showing CRISPR-edited T cells works without serious side-effects. Researchers say that the CRISPR technologies have developed rapidly since the two studies were initiated, and have paved the way for improving the CRISPR gene editing strategies.

First FDA-approved CRISPR application
Our last highlight is the first CRISPR application approved by the FDA. The honour does not go to a CRISPR therapy but to a CRISPR-based diagnostic for SARS-CoV-2 by Sherlock Biosciences.
In hindsight, it is perhaps not surprising with the COVID19 pandemic dominating most 2020, but back in February when Sherlock's CEO, Rahul Dhanda, decided to pivot his company to focus on SARS-CoV-2 the outbreak was just coming out of Wuhan, China.
With CRISPR-Cas13, Sherlock developed a diagnostic tool to detect the SARS-COV-2 virus rapidly, and just three months later in May, the FDA approved the diagnostic tool kit.
Other CRISPR-based diagnostic tools are also being developed (for example fast lab-on-chip, massive surveillance and the graphene-based CRISPR-chip). CRISPR may soon open the door to a new massive surveillance system of emerging infectious diseases. The ability to rapidly and precisely determine circulating viruses will generate data that epidemiologists today can only dream of and help identify future epidemics and stop their spread before developing into global pandemics.
Happy New Year 2021
Medicine is entering a new age of CRISPR gene editing here at CRISPR Medicine News we can hardly wait. We wish for 2021 to be another excellent year for genome editing with tons of new and exciting advances.
Tags
AnalysisArticleBeta ThalassemiaCancerCOVID-19Sickle Cell Disease, SCDCRISPR-CasCas13Cas9
CLINICAL TRIALS
Sponsors:
National Institute of Allergy and Infectious Diseases (NIAID)